This guide clarifies how different honeys stack up for antioxidant potency and what that means for buyers and cooks.
We summarize study results from Manuka, Acacia (Germany), Wild Carrot (Algeria), Brazilian monoflorals (Aroeira, Assa-peixe), and Mt. Olympus samples. You will learn how researchers measure potency — TPC, DPPH/ABTS scavenging, FRAP, and EC50 — and why phenolic content often predicts stronger activity.
Data show Manuka with very high total phenolic content and strong radical-scavenging. Some Mt. Olympus samples match or exceed those assays, and Brazilian monoflorals can approach Manuka’s benchmarks. Results vary with floral source, peroxide activity, and protein factors.
Expect clear explanations of measurement methods, head-to-head comparisons, practical takeaways for diet and cooking, and evidence-based buying tips that reflect botanical origin and lab findings.
Key Takeaways
- Higher total phenolic content usually links to stronger antioxidant activity.
- Manuka often ranks high, but some regional samples can match or exceed it.
- Assays like DPPH, ABTS, FRAP and EC50 help standardize comparisons.
- Composition and origin cause exceptions; look beyond color or price.
- Honey can add dietary antioxidants but works best within a balanced diet.
User intent and what “comparative antioxidant levels in global honey types” really means
Consumers researching honey often want a clear ranking of which varieties deliver the most measurable antioxidant capacity. They ask: how potent is a jar, what health links exist, and which option fits daily meals.
Comparative here means using consistent lab methods—TPC, DPPH/ABTS, FRAP—to rank potency and interpret how those assays reflect kitchen and dietary use.
Chemical composition varies by floral source, geography, processing, storage, and bee species. The same label can show different results depending on harvest and handling.
- Look for reported TPC or DPPH/ABTS values if available.
- Darker jars often indicate higher measured capacity and mineral content.
- Authenticity and gentle processing preserve phenolic content and activity.
Human studies found short-term rises in plasma total phenolics and reducing capacity after honey intake, supporting bioavailability.
| Signal | What it suggests | Action |
|---|---|---|
| Darker color | Higher phenolic content | Prefer for food uses seeking extra antioxidants |
| Listed TPC/FRAP | Quantified activity | Use as selection metric when present |
| Raw / minimal processing | Less loss of active compounds | Choose raw for cooking and finishing |
| Trusted origin | Consistent composition across samples | Buy from reliable producers |
We will map common market honeys to reported capacity and composition so you can pick confidently without wading through dense lab papers.
How antioxidant capacity is measured in honey
Researchers use a set of standardized tests to quantify what honey samples can neutralize or reduce.
Total phenolic content (TPC) is the baseline metric. Labs apply the Folin-Ciocalteu method, read absorbance at 765 nm, and report results as mg GAE/kg or mg GAE/100 g from a gallic acid standard curve. This standardization lets different samples be compared on the same scale.
Free radical scavenging: DPPH and ABTS
The DPPH assay measures loss of absorbance at 517 nm and reports SC50 (mg/mL); lower SC50 = stronger scavenging. ABTS reads at ~734 nm and often captures both hydrophilic and lipophilic activity, so values can differ for the same sample. Trolox or BHT are common reference reagents for calibration.
FRAP and reducing power
FRAP tracks Fe3+ to Fe2+ reduction (absorbance ~700 nm) and reflects reductones and electron-donating capacity. Studies show strong correlations between total phenolic and DPPH/FRAP results, but matrix effects can alter outcomes.
| Assay | Readout / unit | Wavelength (nm) | What it indicates |
|---|---|---|---|
| Folin-Ciocalteu (TPC) | mg GAE/kg or mg GAE/100 g | 765 | Phenolic content standardized to gallic acid |
| DPPH | SC50 (mg/mL) | 517 | Free radical scavenging; lower SC50 = stronger |
| ABTS | SC50 or TEAC | 734 / 730 | Hydrophilic + lipophilic radical neutralization |
| FRAP | µmol Fe2+ equiv./g or absorbance | 700 | Reducing power linked to reductones |
- Why multiple assays matter: each method probes different mechanisms, so combined analysis gives a fuller profile.
- Units and standards: convert mg GAE/kg vs mg GAE/100 g and note SC50 units when comparing values.
- Sources of variance: extraction solvent, incubation time, and optical settings affect reported numbers.
Comparative antioxidant levels in global honey types
A head-to-head look reveals how Manuka, Acacia and Wild Carrot compare across standardized assays and real sample values. Below we normalize reported metrics so readers can scan practical differences quickly.
Head-to-head: Manuka vs. Acacia vs. Wild Carrot
Manuka leads on total phenolic concentration (≈899.09 mg GAE/kg) and shows consistently strong DPPH and ABTS scavenging across studies.
Acacia (Germany) matched or slightly exceeded Manuka on FRAP, indicating stronger reducing power despite similar radical-scavenging profiles.
Wild Carrot (Algeria) registered far weaker activity: DPPH SC50 ~53.31 mg/mL and ABTS ~202.26 mg/mL, a clear contrast with the reference samples.
Regional standouts: Brazil and Mt. Olympus
Brazilian monofloral samples (Aroeira, Assa-peixe) reached TPC near 107.93 mg GAE/100 g and DPPH EC50 values that approach Manuka (A6 11.30 vs. 8.20 mg/mL).
Several Mt. Olympus samples exceeded Manuka on DPPH/ABTS despite TPC clustering from 0.55–0.92 mg GAE/g (Manuka ≈0.71 mg/g). This suggests composition and hydrogen peroxide/protein factors boost activity.
- Quick take: Manuka often tops phenolic counts; Acacia can lead FRAP; Wild Carrot shows low scavenging.
- Regional champions (Brazil, Mt. Olympus) may match Manuka depending on compound profile and assay conditions.
- Practical note: compare units and assay methods before interpreting sample results for food or wellness use.
Results depend on both concentration and composition; assay alignment matters when reading cross-study values.
| Sample | Representative metric | Notable finding |
|---|---|---|
| Manuka (NZ) | TPC ≈899 mg GAE/kg | High phenolics; strong DPPH/ABTS |
| Acacia (Germany) | FRAP > Manuka | Stronger reducing power |
| Wild Carrot (Algeria) | DPPH SC50 53.31 mg/mL | Lower scavenging activity |
| Brazilian monoflorals | TPC up to 107.93 mg GAE/100 g | DPPH EC50 near Manuka |
| Mt. Olympus (Greece) | TPC 0.55–0.92 mg GAE/g | Some samples outperformed Manuka on DPPH/ABTS |
Manuka, Acacia, and Wild Carrot: what the numbers show
When you read the lab results side by side, Manuka, Acacia, and Wild Carrot show distinct assay signatures.
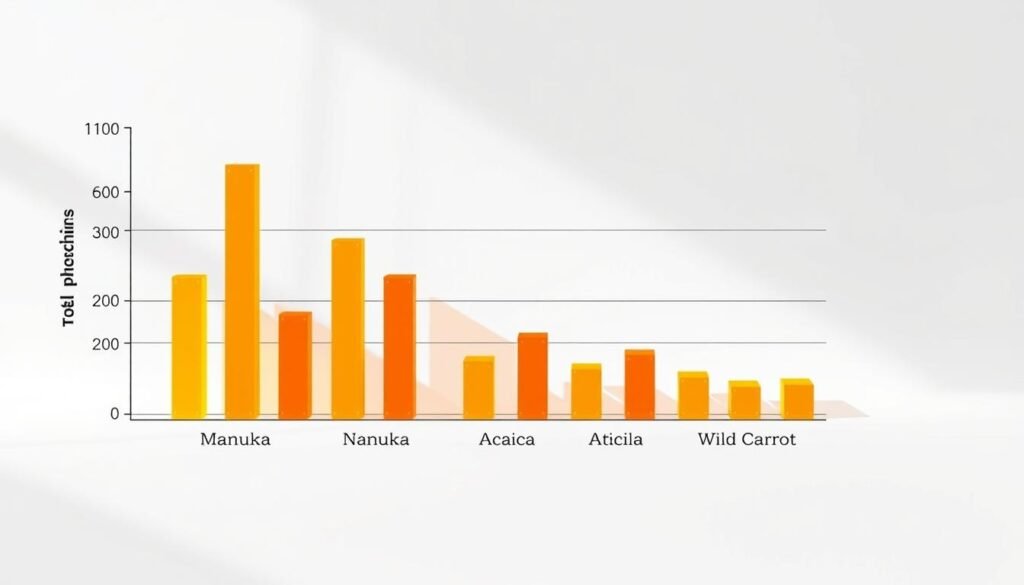
Manuka: phenolic lead and strong scavenging
Manuka (New Zealand) reports total phenolic content around 899.09 mg GAE/kg (gallic acid equivalents).
Its DPPH SC50 is 13.46 mg/mL and ABTS SC50 43.25 mg/mL, showing robust free radical neutralization under standardized assay conditions.
Acacia: higher reducing power
Acacia records lower phenolic content (~627.56 mg GAE/kg) but a slightly higher FRAP (~1.366 ABS700).
Similar DPPH/ABTS values (13.62 and 44.37 mg/mL) place Acacia close to Manuka for radical scavenging while exceeding it on electron-donating activity.
Wild Carrot: lower across assays
Wild Carrot shows reduced activity: TPC ≈503.09 mg GAE/kg, DPPH SC50 53.31 mg/mL, ABTS 202.26 mg/mL, FRAP 0.6386 ABS700.
These values indicate weaker free radical and reducing performance versus the other two sources.
“Lower SC50 values denote stronger radical scavenging; FRAP highlights reducing compounds not counted by total phenolic mass.”
| Sample | TPC (mg GAE/kg) | DPPH SC50 (mg/mL) | FRAP (ABS700) |
|---|---|---|---|
| Manuka (New Zealand) | 899.09 | 13.46 | 1.2106 |
| Acacia | 627.56 | 13.62 | 1.366 |
| Wild Carrot | 503.09 | 53.31 | 0.6386 |
Takeaway: use both total phenolic content and specific assay results when choosing a honey for higher antioxidant activity. Composition and source profile shape the final performance.
Brazilian monofloral honeys: phenolics, flavonoids, and DPPH EC50 compared to Manuka
Brazilian monofloral samples from Minas Gerais show a broad TPC range but include top performers that approach premium benchmarks.
Aroeira and Assa-peixe: phenolic and flavonoid content
Top Aroeira and Assa-peixe specimens reached TPC near 107.93 mg GAE/100 g, signaling substantial phenolic presence.
Flavonoids for A1-Aroeira measured about 18.94 mg QE/100 g and 7.00 mg RE/100 g. Manuka (MGO 30+) recorded 20.43 mg QE/100 g and 7.58 mg RE/100 g, so some Brazilian contents closely match the reference.
DPPH EC50 and scavenging efficiency
DPPH EC50 gives a direct view of radical-scavenging efficiency. A6-Aroeira’s EC50 at 11.30 mg/mL sits near Manuka’s 8.20 mg/mL, indicating competitive activity for select samples.
Many of these honey samples were dark amber. That color often correlates with higher measured action and can serve as a quick visual cue for shoppers.
- Select samples approach Manuka on flavonoid and TPC metrics.
- EC50 comparisons show Aroeira can be close in radical-scavenging performance.
- Botanical authentication (melissopalynology) helps ensure consistent composition across jars.
| Sample | TPC (mg GAE/100 g) | Flavonoids (QE/RE mg/100 g) | DPPH EC50 (mg/mL) |
|---|---|---|---|
| Aroeira (top) | ~107.93 | 18.94 / 7.00 | 11.30 |
| Assa-peixe (representative) | ~95.00 | ~17.50 / 6.50 | ~12.50 |
| Manuka (MGO 30+) | 141.73 | 20.43 / 7.58 | 8.20 |
Takeaway: while average TPC for these Brazilian honeys trails Manuka, select monofloral samples deliver meaningful potency and favorable flavonoid profiles. Look for verified botanical source and lab-reported TPC or EC50 values when choosing jars for higher antioxidant activity.
Mt. Olympus honeys: when polyphenol quality outperforms quantity
Several Mt. Olympus jars outperformed high-MGO New Zealand controls on DPPH and ABTS scavenging.
Greek samples from this biodiversity-rich region showed strong radical-scavenging even though measured total phenolic values clustered near 0.55–0.92 mg GAE/g. Manuka used as a New Zealand control sat at about 0.71 mg/g, yet some Greek specimens returned lower IC50s on dpph and ABTS assays.
Why this happens: specific phenolic profiles and matrix factors can yield higher measured activity than raw phenolic mass suggests. Tests also flagged a role for hydrogen peroxide and proteinaceous compounds. MIC shifts after catalase and proteinase K treatment support this mechanism.
Freeze-dried powders kept most activity despite reduced total phenolic readings, which points to compound stability and quality over simple quantity.
- Assay differences reveal distinct mechanisms behind measured activity.
- Floral source within Mt. Olympus affects composition and predictability.
- Consider regional jars with lab data when seeking potent antioxidant options.
| Feature | Mt. Olympus finding | Implication |
|---|---|---|
| Total phenolic (mg GAE/g) | 0.55–0.92 | Comparable to controls |
| DPPH / ABTS | Some IC50s lower than New Zealand Manuka | Higher radical-scavenging activity |
| Auxiliary factors | Hydrogen peroxide & proteinaceous compounds | Contribute to antibacterial and reducing action |
From chemistry to benefits: why phenolic content correlates with antioxidant activity
The chemical mix inside a jar — from flavonoids to melanoidins — shapes how well it reduces oxidative stress in lab tests.
Phenolic acids and flavonoids act as primary radical scavengers. They donate electrons and stabilize free radicals, which lowers measured DPPH and FRAP signals.
Ascorbic acid and certain amino acids add reducing power. Maillard-derived melanoidins and carotenoid derivatives also contribute sustained activity.
Enzymes such as glucose oxidase generate low levels of hydrogen peroxide, while catalase modulates that signal. Amino acids and proteins can participate in redox reactions, altering assay outcomes.
How composition maps to biological effect
Studies show higher total phenolic content usually links to stronger DPPH/FRAP results and to protective effects in cell models. Phenolic fractions reduced oxidative markers in endothelial assays, tying chemistry to potential health relevance.
Composition matters: two jars with similar total phenolic content can differ if one has more potent flavonoids. Synergy among compounds often produces greater activity than any single molecule alone.
Practical note: TPC is a useful predictor, but a full profile (flavonoid specifics, enzyme activity, melanoidins) gives a clearer picture for food uses and dietary benefit.
| Compound class | Primary role | Assay impact | Practical implication |
|---|---|---|---|
| Phenolic acids / flavonoids | Electron donation, radical stabilization | Strong DPPH and FRAP correlation | Look for high TPC / flavonoid reports |
| Ascorbic acid / amino acids | Reducing agents | Boosts reducing power (FRAP) | Enhances short-term protection |
| Melanoidins / carotenoids | Maillard and pigment-derived activity | Contributes to sustained scavenging | Often linked to darker color and shelf stability |
| Enzymes (glucose oxidase/catalase) | Peroxide production / modulation | Alters observed antimicrobial and redox signals | Matrix effects; processing changes outcomes |
Free radicals, hydrogen peroxide, and antioxidant mechanisms in honey
Lab assays separate how honey neutralizes radicals from how it donates electrons, revealing distinct protective routes.
Scavenging radicals vs. reducing metal ions
DPPH (517 nm) and ABTS (730–734 nm) directly measure how well a sample captures a free radical or radical cation. ABTS detects both water- and fat-soluble molecules, so results can differ from DPPH for the same jar.
FRAP measures electron transfer by following Fe3+ reduction to Fe2+. That readout reflects reductones and other electron-donating compounds rather than direct radical capture.
Some samples show extra apparent capacity because glucose oxidase generates hydrogen peroxide. That peroxide engages in redox chemistry and can boost measured activity alongside phenolic compounds.
Organic acids and specific phenolic acids also shift pH and reaction kinetics. Matrix components may stabilize reactive species or help regenerate antioxidants, extending measured activity during assays.
“Multiple assays together give a fuller, more reliable profile than any single test.”
- Different assays probe complementary mechanisms and should be read together.
- Controls and replication help separate enzymatic peroxide effects from non-enzymatic scavenging.
- Use multi-assay panels to guide practical choices for cooking or supplements.
| Mechanism | Assay example | What it shows |
|---|---|---|
| Radical scavenging | DPPH / ABTS | Direct neutralization of radicals; ABTS is more inclusive of hydrophilic/lipophilic compounds |
| Reducing power | FRAP | Electron donation to reduce Fe3+ to Fe2+; indicates reductones |
| Enzymatic peroxide contribution | Catalase-controlled assays | Shows hydrogen peroxide-driven effects separate from phenolic scavenging |
What darker color often signals: antioxidant properties and mineral influence
Dark amber jars often point to richer phenolic pigments and higher measured activity. Color reflects the botanical source, climate during nectar flow, hive temperature, and mineral uptake. These factors shape the final composition and observable hue.
Darker color frequently correlates with greater phenolic content and pigment classes such as anthocyanins and flavones. Minerals and certain acids also shift color while supporting redox reactions that show up in assays.
Processing and storage can deepen color through Maillard reactions, creating melanoidins that add reducing power. That means aged or gently heated jars may test higher even if raw content was similar.
Use color as a practical cue, not a guarantee. When antioxidants matter, pair visual checks with reported TPC or assay values. For more on phenolic content and measured activity, see this phenolic content research.
- Tip: darker jars often bring bolder flavor and suit savory or robust food pairings.
- Note: lab data remains the most reliable guide for true activity and values.
Why origin matters: botanical source, geography, bee species, and processing
Origin shapes what a jar delivers on taste, stability, and measured activity. Botanical source sets the baseline mix of phenolic compounds, enzymes, and minerals that drive assay results. A flower’s nectar determines which compounds bees collect, so monofloral jars often show consistent profiles when authenticated by pollen analysis.

How floral profile, climate, and storage shift phenolic content and activity
Floral source: Different floral sources yield distinct phenolic fingerprints and enzyme complements. These compounds directly affect common lab readouts such as TPC, DPPH/ABTS, and FRAP.
Geography and season: Soil minerals, rainfall, and seasonal temperature alter nectar chemistry. Drier seasons can concentrate sugars and some phenolics, while heavy rains dilute nectar and lower measured contents.
Bee species and hive conditions: Native bee strains and hive management change enzyme input and moisture. Glucose oxidase production and water content affect both activity and shelf life.
Processing and storage: Heat, light, and time break down sensitive flavonoids and enzymes or transform them into Maillard products. Careful handling preserves lab-measured performance; poor storage skews results downward.
“Melissopalynology and transparent sourcing let buyers predict composition and expect more reliable assay results.”
| Factor | How it changes composition | Practical implication |
|---|---|---|
| Botanical source | Alters phenolic profile and enzyme mix | Choose authenticated monofloral samples for consistency |
| Geography / climate | Shifts nectar chemistry and mineral content | Regional jars can outperform labels from other zones |
| Bee species / hive | Modulates enzymatic contributions and moisture | Look for producer notes on apiary practices |
| Processing / storage | Degrades or transforms compounds over time | Prefer raw, gently handled jars with provenance details |
- Study support: Multiple studies report significant differences among samples from different floral profiles on TPC, DPPH/ABTS, and FRAP metrics.
- Buyer advice: Prioritize jars with source transparency and verified assay data for predictable results.
Methodology nuances that affect reported values
How labs run their tests — solvents, temperature, and calibration — often explains discordant results more than regional origin. Small changes in protocol can shift reported activity and total phenolic numbers enough to alter conclusions about a jar.
Key protocol factors
Wavelengths and units matter. Folin–Ciocalteu readings use 725–765 nm and report mg GAE/kg or mg GAE/100 g, so convert units before direct comparison.
DPPH and ABTS assays use methanolic solutions with Trolox or BHT as calibration standards. SC50/IC50 are given as mg sample/mL; FRAP reads at ~700 nm to show reducing power.
What changes measured activity
- Solvent strength (e.g., 50% methanol vs. pure methanol) and extraction method shift which acids and flavonoids are pulled from samples.
- Incubation time and temperature alter reaction kinetics and final values.
- ABTS detects both hydrophilic and lipophilic antioxidants, often producing higher SC50 than DPPH for the same samples.
- Endogenous peroxide or enzyme activity can inflate apparent results unless catalase/proteinase controls are applied.
Best practices for reliable analysis
Replication, blanks, and clear standards are essential. Run triplicates, include reagent blanks, and state calibration agents like Trolox or BHT.
“Read methods and units carefully; method details explain much of the variation between published results.”
| Factor | Effect on results | Practical tip |
|---|---|---|
| Extraction solvent | Changes which compounds are measured | Compare studies using the same solvent |
| Calibration standard | Benchmarks scavenging across assays | Note Trolox/BHT when comparing SC50 values |
| Enzymes / peroxide | Can inflate apparent activity | Prefer studies that report catalase controls |
Bottom line: standardized methods and transparent reporting let consumers, labs, and food professionals make fair comparisons of antioxidant activity and select samples based on reliable results.
Health relevance in the real world: what human data suggest
Clinical trials report quick rises in plasma phenolic markers after modest servings of jarred nectar.
Short-term increases in plasma total phenolics and antioxidant capacity after intake
Controlled studies showed that single doses can acutely raise plasma total phenolics and measured antioxidant capacity within hours.
Daily intake produced measurable results: blood ascorbic acid rose by 47%, β-carotene by 3%, uric acid by 12%, and glutathione reductase activity by 7% in reported trials.
Cell studies align with human data. Phenolic-rich fractions protected endothelial cells from oxidative stress and reduced markers of free radical damage.
“These findings indicate bioavailability of active compounds under typical food use.”
Practical points: benefits vary by floral source because phenolic and flavonoid profiles differ. Choose jars with higher reported TPC and strong assay activity when seeking dietary support.
Moderation matters. Integrate this natural sweetener as part of balanced food choices, mindful of sugar and caloric effects. More long-term studies are needed to quantify sustained health impacts.
| Evidence type | Reported change | Implication |
|---|---|---|
| Short-term human trials | ↑ plasma phenolics; ↑ antioxidant capacity | Acute bioavailability after normal servings |
| Daily intake studies | ↑ vitamin C, β-carotene, uric acid, enzyme activity | Improved micronutrient and endogenous defenses |
| Endothelial cell assays | Reduced oxidative damage | Phenolic/flavonoid fractions show protective properties |
Practical comparison guide: choosing honey for antioxidant capacity
Picking a jar for higher antioxidant potential need not be guesswork. Focus on measurable signals that labs report and practical cues you can check on the label or by sight.
Signals to look for: TPC/GAE, DPPH/ABTS, darker color, and trusted origin
Look for lab-reported total phenolic content (mg GAE per unit). That value gives a quick estimate of phenolic-rich samples and helps compare different samples objectively.
Prefer published DPPH or ABTS results showing lower EC50/IC50. Lower values mean more efficient free radical scavenging under assay conditions.
- Darker color often aligns with higher activity; use it as a visual cue, then verify with lab metrics.
- Trusted origin disclosures (botanical source, region, harvest date) and third-party checks improve consistency across jars.
- Flavonoid or specific phenolic profiles, when available, help explain potency beyond a single total phenolic figure.
“Align reported units and methods before you compare values across different sources.”
| Signal | What it suggests | Action |
|---|---|---|
| Total phenolic content (mg GAE) | Higher phenolic mass | Use as primary selection metric |
| DPPH/ABTS (EC50/IC50) | Free radical efficiency | Prefer lower values for stronger scavenging |
| Origin / melissopalynology | Composition consistency | Choose authenticated monofloral or vetted regional jars |
Practical tips: For food use, pair robust, darker jars with bold recipes—teas, marinades, and roasted dishes benefit most. Rotate a verified high-TPC monofloral with a strong regional sample to diversify flavonoid intake. For verification and grading context, consult published analyses such as this research on phenolic content (phenolic content study) and a review of grading standards (honey grading standards).
New Zealand’s Manuka vs. regional champions: who leads by metric
Different metrics crown different leaders—Manuka for phenolic content, and select regional jars for scavenging performance.
Phenolic content leaders vs. DPPH/ABTS scavenging leaders
Manuka remains the clear phenolic leader by mass (≈899 mg GAE/kg) and shows strong DPPH/ABTS activity across many published results.
Yet some Mt. Olympus samples returned lower IC50s on DPPH and ABTS, indicating superior radical-scavenging despite lower total phenolics. Catalase and protease tests suggest that peroxide and proteinaceous compounds boost measured activity in those Greek jars.
Brazilian Aroeira narrows the gap: select honey samples reported DPPH EC50 values near Manuka’s benchmark, making Aroeira a near-leader for real-world scavenging activity.
Acacia shows the opposite pattern: moderate TPC but higher FRAP, so it leads on reducing power rather than phenolic mass or radical capture.
“Pick the metric that matters: TPC for phenolics, DPPH/ABTS for radical-scavenging, FRAP for reducing power.”
- Compare values within the same assay framework to ensure fair interpretation.
- Use one jar for high phenolic content and another known for top scavenging to cover multiple activity routes.
- Ongoing assay work will refine leaderboards as more honey samples are tested.
| Leader | Metric | Notable result |
|---|---|---|
| Manuka (New Zealand) | Total phenolic content | ≈899 mg GAE/kg; strong DPPH/ABTS baseline |
| Mt. Olympus (Greece) | DPPH / ABTS scavenging | Some IC50s lower than Manuka; peroxide/protein contributions |
| Aroeira (Brazil) | DPPH EC50 | Selected samples approach Manuka’s EC50 values |
| Acacia (Germany) | FRAP (reducing power) | Higher FRAP despite moderate TPC |
Bottom line: New Zealand Manuka remains a reliable high performer, but regional champions can compete or excel depending on the assay and mechanism—phenolics, peroxide, or protein-driven pathways.
Limits and caveats: variability, assay differences, and quality control
Reported values depend heavily on how a study runs its assays and reports units. Small changes in extraction solvent, incubation time, or calibration standard shift results enough to change rankings.
ABTS often reads differently from DPPH because it captures both hydrophilic and lipophilic activity. Catalase and proteinase K treatments show that endogenous peroxide or protein fragments can inflate apparent activity unless controlled.
Biological variability matters: two jars labeled the same can show different contents across seasons, floral mix, or microclimates. That makes single-sample claims weak when applied to broad purchase decisions.
Quality control and transparent methods are essential. Prefer reports from reputable labs that state units, standards, and replication details.
“Unit conversions and method alignment are essential before comparing reported values.”
- Values are not universally interchangeable; align units and method before comparison.
- Request authenticated origin and batch-level testing for reliable selection.
- Replicate tests and cross-assay confirmation improve confidence for any lot or product.
- Storage and handling degrade antioxidants over time; treat measured activity as time-sensitive.
| Issue | Impact | Action |
|---|---|---|
| Method variance | Shifts reported values | Compare like-for-like assays |
| Enzymatic peroxide | Inflates activity | Check catalase-controlled analysis |
| Sample variability | Different contents across batches | Favor authenticated, replicated studies |
Consumer takeaway: treat single-study claims cautiously. Look for transparent lab analysis, clear units, and consistent testing across multiple samples before using results to guide food purchases or health choices.
Conclusion
Across assays, no single source wins every category—Manuka excels on total phenolics while some regional jars outperform for scavenging or reducing power.
This review shows Manuka often leads by phenolic content, Acacia stands out on FRAP, and Wild Carrot scores lower on multiple tests. Select Brazilian and Mt. Olympus samples can match or surpass specific metrics.
Multiple compounds — phenolic acids, flavonoids, ascorbic acid and amino acids — work together to create measurable antioxidant activity. Origin and processing shape those properties, so strong, source transparency and verified lab data matter for buyers.
For practical picks, favor higher TPC/GAE, low DPPH/ABTS values and darker jars as an initial cue, then confirm with reported assay results. Human studies support short-term rises in antioxidant capacity after normal servings. Try vetted honeys to balance flavor and function, and support ongoing standardization for clearer comparisons.
FAQ
What does "comparative antioxidant levels in global honey types" mean for a consumer?
It refers to measuring and comparing compounds like total phenolic content (TPC), flavonoids, and ascorbic acid across honeys from different regions. These measures — often reported as gallic acid equivalents (GAE) or via assays such as DPPH, ABTS, and FRAP — indicate the capacity of a honey sample to neutralize free radicals or reduce oxidants like hydrogen peroxide. For shoppers, it helps identify honey with higher antioxidant capacity and potential added health value.
How is antioxidant capacity of honey measured?
Labs use several standard methods. TPC is often determined by the Folin–Ciocalteu method and expressed as mg GAE per kg or per 100 g. Radical-scavenging assays like DPPH and ABTS test the honey’s ability to neutralize specific free radicals; results may be shown as EC50 or percent inhibition. FRAP measures ferric reducing antioxidant power, reflecting reducing agents such as reductones, phenolic acids, and certain proteins.
Which honey types typically show the highest TPC and radical-scavenging activity?
Certain monofloral varieties stand out. New Zealand Manuka often reports very high TPC (reported near 899 mg GAE/kg in some studies) and strong DPPH/ABTS profiles. Some Brazilian monoflorals like Aroeira and Assa-peixe also show high phenolic and flavonoid contents. Greek samples from Mt. Olympus have, in some cases, matched or exceeded Manuka in DPPH/ABTS despite similar TPC.
Is darker honey always higher in antioxidant compounds?
Darker color often correlates with higher mineral content, melanoidins, and phenolics, which can boost antioxidant capacity. However, color alone is not definitive. Chemical composition, floral source, and specific phenolic profiles better predict activity than color by itself.
How do Manuka, Acacia, and Wild Carrot compare chemically?
Manuka typically shows the highest total phenolic content and strong radical-scavenging activity. Acacia often has moderate phenolics but can show comparable FRAP values and consistent scavenging performance. Wild Carrot samples generally exhibit lower phenolics and weaker activity across assays compared with the other two.
What role do phenolic acids and flavonoids play in honey’s activity?
Phenolic acids and flavonoids donate electrons or hydrogen atoms to neutralize free radicals, contributing directly to DPPH/ABTS scavenging and FRAP results. They work alongside other compounds — ascorbic acid, amino acids, proteins, hydrogen peroxide, and melanoidins — to shape overall antioxidant capacity.
Can honey increase antioxidant measures in humans after consumption?
Short-term studies report transient rises in plasma total phenolics and overall antioxidant capacity after ingesting certain honeys. Effects vary by dose, honey type, and individual metabolism. These acute changes do not equate to long-term clinical outcomes without further evidence.
Why do reported values vary so much between studies?
Variability stems from differences in assay protocols, reagents and standards (Trolox, BHT, gallic acid), sample preparation, reporting units (mg/kg vs. mg/100 g), and natural variability: floral profile, geographic origin, bee species, and storage. Small methodological shifts can change reported TPC, DPPH EC50, ABTS percent inhibition, and FRAP results.
How should I choose honey if I want higher antioxidant capacity?
Look for independent lab data or labels that report TPC (GAE), DPPH/ABTS results, or FRAP values. Prefer reputable origin information and darker monofloral honeys known for higher phenolics, such as Manuka or certain Brazilian monoflorals. Verify minimal processing and proper storage to preserve phenolics and peroxide activity.
Are regional honeys like Brazilian or Greek samples competitive with Manuka?
Yes. Some Brazilian monoflorals (Aroeira, Assa-peixe) report TPC and flavonoid contents approaching Manuka, and certain Mt. Olympus Greek honeys have outperformed Manuka in DPPH/ABTS assays due to qualitatively potent polyphenols and contributory factors like hydrogen peroxide and proteinaceous compounds.
What practical signals on a label indicate stronger activity?
Useful indicators include stated TPC (GAE), mention of DPPH/ABTS or FRAP testing, floral source (monofloral names), country or region of origin, and minimal processing claims. Certifications or third‑party lab reports add confidence in reported antioxidant capacity.
Do peroxide and non-peroxide factors both matter?
Yes. Hydrogen peroxide produced enzymatically in some honeys contributes to antimicrobial and oxidative effects. Non‑peroxide components — phenolics, flavonoids, ascorbic acid, amino acids, melanoidins, and proteins — drive radical-scavenging and reducing activity measured by DPPH, ABTS, and FRAP.




