Controlled mating offers bee managers a reliable path to meet selection goals and improve stock. Artificial pairing replaces random flight mating and gives clear data for genetics and research.
Why this matters: a honey bee monarch often mates with many drones during brief flights, which mixes sperm and makes predictable selection hard. Using a precise mating method lets queen producers and beekeepers choose drone sources, set the number of drones represented, and control semen dose.
Standard dose per female is about 8–10 microliters. Success depends on careful technique, good handling, and strong colony care. When those elements are in place, artificially mated queens can match or exceed natural peers in laying and longevity.
Practical impact: this approach supports rigorous selection of traits, yields repeatable results for research, and improves program control for U.S. beekeepers focused on honey and colony performance.
Key Takeaways
- Artificial mating gives precise control over drone sources and dose.
- Natural mating is highly random; control improves selection outcomes.
- Typical semen dose is 8–10 microliters per queen.
- Good technique and post-care are essential for success.
- Producers gain clearer parentage links and better program data.
Why controlled mating matters now in U.S. queen breeding programs
Predictable pairings are becoming essential for U.S. bee programs facing Varroa and genetic drift. Controlled mating gives producers a repeatable way to protect temperament, honey yield, and winter survival while advancing selection for mite tolerance and other key traits.
Breeding objectives: balance and priorities
Queen producers and beekeepers must set weighted goals. Prioritize Varroa tolerance, consistent honey production, calm behavior, and reliable overwintering.
Keep records that link parents to colony outcomes. That data makes genetics actionable across cycles.
When to choose controlled mating versus natural mating
Use instrumental insemination when you need fixed drone sources, exact semen doses, or specific crosses for research and tight selection.
Rely on natural mating when local adaptation and scale are more important and drone saturation yards are available.
Readiness checklist for program control
- Confirm selected stock and access to mature drones (mature at ~2 weeks, peak ~3 weeks).
- Invest in precision tools, CO2, lighting, and training time.
- Plan for rearing capacity, post-procedure follow-up, and strict recordkeeping.
“Controlled mating reduces the chance of unwanted genetics entering your program and safeguards gentle behavior around people and livestock.”
Instrumental insemination in queen breeding: a step-by-step how-to
Good equipment, careful handling, and clean technique turn a complex task into a predictable workflow.

Essential equipment and setup
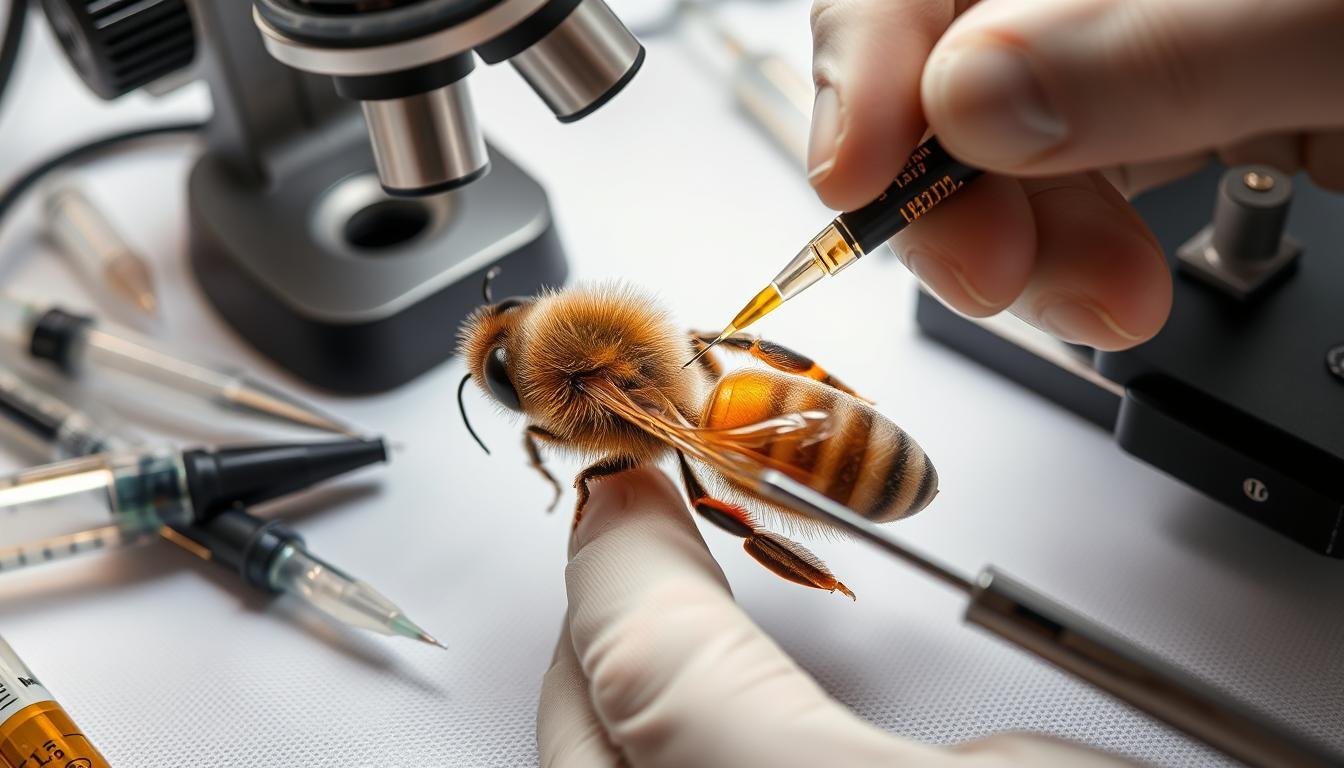
Bench: rigid stand with micro-manipulators, a 10x–20x microscope, and a cool LED light to avoid tissue drying. Use a CO2 source with a flow regulator at the queen holder for controlled anesthesia.
Preparing live material
Rear vigorous virgins and keep donor colonies strong. Drones mature ~2 weeks after emergence and peak near week 3. Plan overproduction so you can cull weak males.
Semen collection and handling
Expect about 1 μL per drone and roughly half to yield usable semen. Use a Harbo syringe with compatible tips and a Schley manipulator for repeatable control. Reject any sample with feces or mucus.
Insemination procedure and post-care
Position the queen, open the sting chamber, bypass the valvefold, and deliver a precise 8–10 μL dose. Bank queens ~2 days to allow sperm migration. Track outcomes and dissect failed cases to refine the process.
“Skill builds with planned practice; aim for consistent records and a runner to keep throughput steady.”
Optimizing success: equipment choices, sanitation, genetics, and advanced applications
Precision tools, careful sanitation, and clear goals separate routine practice from program-level success.
Choosing and maintaining instruments
Select stands and micro-manipulators that allow steady, repeatable motion. Confirm compatibility among holders, syringe tips, and microscope adapters because parts are not standardized. Many teams use a Schley rig with a Harbo syringe for reliable semen collection and dosing.
Sanitation and common pitfalls
Disinfect tools between uses, guard against drone feces during eversion, and use cool LED lighting to avoid heat stress. Minimize handling time to reduce injury and infection risk.
Compare mated queens and naturally mated cohorts by brood patterns, laying continuity, and longevity. Store semen at room temperature for short-term use (days to weeks); avoid refrigeration. For long-term needs, cryopreservation supports conservation and research but may alter laying patterns.
Design and program excellence
Use single-drone crosses to isolate traits or pooled semen to raise effective population size. Invest in training, years of practice, and strict records. Consistent technique, disciplined rearing schedules, and good data let beekeepers and breeders turn technique into production gains.
“Treat semen as a critical reagent and your instruments as precision lab tools.”
Conclusion
, Controlled mating offers a practical path to repeatable genetics and better honey production across U.S. apiaries. Use precise methods with good equipment, strict sanitation, mature drones, and careful queen handling to deliver consistent, high-performing mated queens.
Plan semen doses at 8–10 μL, use short-term room-temperature storage for flexible scheduling, and consider cryopreservation for long-term stock and research needs. Accept limits—drone availability and clean collection—but mitigate them with rearing plans and workflow support.
Commit to training, standards, and multi-year records so beekeepers and breeders can compare instrumental insemination and natural mating cohorts, confirm trait gains, and turn controlled mating into a durable tool for stronger colonies and reliable production.
FAQ
What role does controlled mating play in modern U.S. queen programs?
Controlled mating gives breeders precise genetic direction. It helps balance traits such as Varroa tolerance, honey production, docility, and overwintering survival. By using targeted crosses, producers reduce unwanted variability from open mating and accelerate improvement of breeding objectives across apiaries.
How do I decide between controlled mating and natural field mating?
Choose controlled mating when you need predictable outcomes, want to fix specific traits, or run a closed population program. Natural mating suits large-scale commercial producers who prioritize throughput and rely on regional drone availability. Resource limits and goals determine the best method.
What should be on my readiness checklist before starting a controlled mating program?
Ensure you have selected productive breeder stock, access to healthy drones, proper equipment (microscope, precision syringe systems, CO2 source, and focused lighting), trained personnel, and a plan for recordkeeping and colony follow-up. Budget for training and several seasons to reach reliable results.
What essential tools are required for precise mating procedures?
Key tools include a stereomicroscope, a quality syringe or micro-capillary system, a CO2 delivery setup for temporary anesthesia, cold light for clear visualization, and fine manipulators. Compatibility and calibration matter more than brand; maintenance and spare parts are critical.
How do I prepare live material: drones and virgin females?
Rear drones from selected stock and time their emergence so mature males peak during collections. Raise virgins in strong nucleus colonies to ensure vigour. Confirm sexual maturity and handle both sexes gently to avoid injury or stress before processing.
What are best practices for semen collection from drones?
Use clean technique to avoid contamination: collect under a microscope, ensure full eversion for maximal yield, pool when needed for genetic goals, and work quickly. Expect variable yields per drone; plan numbers accordingly for target volume.
Can you summarize the critical steps of the mating procedure itself?
Key steps include proper positioning of the female, careful bypassing of the valvefold to access the reproductive tract, controlled dosage delivery, and minimal handling time. Gentle restraint and steady, precise movements reduce injury and improve success rates.
What post-procedure care improves acceptance and performance?
Use timed CO2 recovery protocols, consider short-term banking in queen cages on host colonies, monitor for normal laying onset, and introduce queens to nucleus colonies under protective measures. Track sperm migration and initial brood patterns to confirm success.
What realistic throughput can a trained technician achieve?
Throughput depends on skill, setup, and support. Beginners will process only a few per day; trained teams can handle many more. Set practical daily targets, document success rates, and scale staff and equipment as proficiency improves.
How should I choose and maintain instrumentation for long-term use?
Select reliable, repairable systems with available parts. Regular calibration, cleaning, and gentle handling extend life. Keep a maintenance log, spare syringes or capillaries, and a tested CO2 regulator to avoid downtime during peak seasons.
What sanitation steps prevent contamination and injury?
Sterilize collection and delivery tools between uses, work in a clean workspace with stable temperature, avoid cross-contamination between batches, and use disposable or easily disinfected surfaces. Proper lighting and magnification reduce accidental trauma.
How do I evaluate outcomes versus naturally mated stock?
Compare laying patterns, brood viability, colony temperament, honey yields, and overwinter survival. Record sperm viability checks and long-term productivity metrics. Controlled matings offer clearer attribution of trait changes to specific crosses.
What are current options for managing semen between collection and use?
Short-term storage at cool, stable temperatures with appropriate diluents suits same-day use. Transport requires insulated containers and quick turnaround. Research on cryopreservation is progressing, but routine long-term frozen storage remains specialized.
How do breeding designs like single-drone crosses or pooled semen affect programs?
Single-drone crosses give precise parentage for experimental or research purposes. Pooled semen increases genetic diversity and eases logistics for production. Choose designs based on selection intensity, desired control over pedigrees, and program scale.
What practices build a high-quality long-term program?
Invest in formal training, expect several years to refine techniques, keep meticulous records, use data to drive selection, and maintain strong governance over stock and disease control. Continuous improvement and collaboration with extension services or universities accelerate progress.